Posts
Kidney Day Nutrition Awareness
March 11, 2026
Post-Holi Health Reset: Simple Diet & Lifestyle Tips:-
March 6, 2026
#Mineral #Rich #Immunity #Diet:-
March 5, 2026
After Holi Detox Diet
March 5, 2026
The Dawn of a New Era: How AI is Revolutionizing Drug Discovery
February 16, 2026
FDA Approves First-of-Its-Kind Device to Treat Pancreatic Cancer
February 16, 2026
The Hidden Gut Bacteria That Could Revolutionize Your Health
February 15, 2026
The Future of Diagnosis: How AI is Revolutionizing Brain Scans
February 14, 2026
Scientists Discover Protein That Can Rejuvenate Aging Brain Cells
February 13, 2026
A New Dawn for Pancreatic Cancer Treatment: FDA Approves First-of-Its-Kind Wearable Device
February 13, 2026
AI Revolutionizes Brain Scan Analysis, Offering Diagnoses in Seconds
February 12, 2026
Managing Flu Season: A Guide for 2026
February 12, 2026
Your Immune System's Molecular Diary: How Life Experiences Shape Your Health
February 11, 2026
The Origami of Life: How DNA Folding and Immune System Secrets are Revolutionizing Medicine
February 10, 2026
Revolutionary AI Technology Reads Brain MRIs in Seconds: A Game-Changer for Neurological Diagnosis
February 10, 2026
A Beautiful Gesture of Gratitude: Akanksha’s Journey to Motherhood at Saksham Hospital
November 17, 2025
Your Guide to Ayushman Bharat at Saksham Hospital, Saharanpur
November 17, 2025
Understanding Nasal Allergy & Turbinate Hypertrophy: Causes, Symptoms & Treatment
November 16, 2025
The Importance of Regular Health Check-ups
October 2, 2025
❤️ Your Heart Matters: A Guide to Better Heart Health
September 30, 2025
A day in the life of an Obstetrician
September 28, 2025
A New Dawn for Pancreatic Cancer Treatment: FDA Approves First-of-Its-Kind Wearable Device
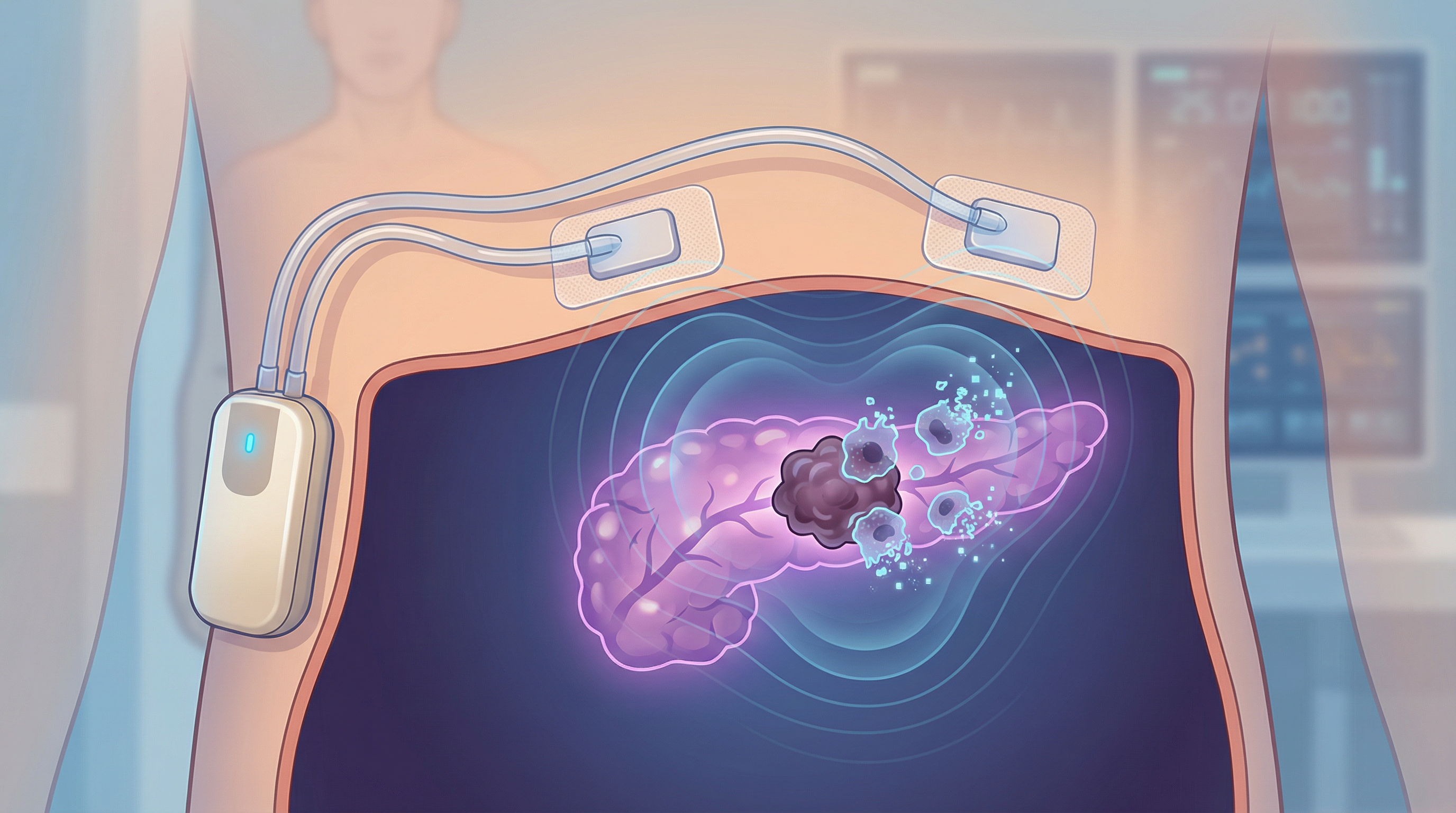
A New Dawn for Pancreatic Cancer Treatment: FDA Approves First-of-Its-Kind Wearable Device
For decades, a diagnosis of pancreatic cancer has been a devastating blow, with treatment options remaining stubbornly limited and survival rates showing only modest improvements. It is one of the most lethal cancers, often diagnosed late and notoriously difficult to treat. But today, a significant breakthrough offers a new beacon of hope. The U.S. Food and Drug Administration (FDA) has approved Optune Pax, a revolutionary wearable device that represents the first new treatment for locally advanced pancreatic cancer in nearly 30 years. This non-invasive, home-based therapy is poised to change the landscape of cancer care, offering patients a new lease on life.
Introducing Optune Pax: A Biophysical Approach to Fighting Cancer
Developed by Novocure, Optune Pax is a portable, non-invasive device that utilizes a groundbreaking technology called Tumor Treating Fields (TTFields). Unlike traditional chemotherapy that uses drugs to kill cancer cells, TTFields employ a biophysical approach. The device delivers low-intensity, alternating electrical fields to the abdomen through a series of wearable adhesive patches. These electrical fields are precisely tuned to disrupt the rapid cell division that is a hallmark of cancer, causing cancer cells to die while leaving healthy cells largely unharmed. This is possible because healthy cells have different electrical properties and divide at a much slower rate than cancerous ones.
The device is designed for continuous use, allowing patients to receive treatment while going about their daily lives. The portable generator is carried in a small bag, offering a level of freedom and convenience that is a world away from traditional, hospital-based cancer therapies. This aligns with the FDA's "Home as a Health Care Hub Initiative," which champions patient-centered devices that seamlessly integrate into people's lives.
The PANOVA-3 Trial: Clinically Proven to Extend Life and Improve Quality of Life
The FDA's approval is based on the results of the international, randomized, and controlled Phase 3 PANOVA-3 clinical trial. The study, which enrolled 571 patients, demonstrated a statistically significant improvement in overall survival for patients treated with Optune Pax in combination with standard chemotherapy (gemcitabine and nab-paclitaxel) compared to chemotherapy alone. Patients receiving the combination therapy saw their median overall survival increase by two months. While this may seem modest, in the context of pancreatic cancer, it is a significant step forward.
Perhaps even more importantly, the trial showed a dramatic improvement in patients' quality of life. A key challenge in pancreatic cancer is managing the debilitating pain that often accompanies the disease. The PANOVA-3 trial revealed that patients treated with Optune Pax had a median time to pain progression of 15.2 months, a remarkable 6.1-month extension compared to the 9.1 months for those on chemotherapy alone. This means patients can live longer, more comfortable lives, with less reliance on pain medication.
A "Practice-Changing" Development for a Challenging Disease
The medical community has hailed the approval of Optune Pax as a landmark achievement. Dr. Vincent Picozzi, a medical oncologist and investigator in the PANOVA-3 trial, called the device "practice-changing," highlighting the improvement in overall survival without adding to the systemic side effects commonly associated with chemotherapy. Frank Leonard, CEO of Novocure, emphasized the novelty of the treatment, stating that "Optune Pax is a fundamentally different treatment, utilizing a biophysical approach that targets the unique electrical properties of cancer cells."
Pancreatic cancer remains a formidable foe. In the U.S. alone, it is the third leading cause of cancer-related deaths, with a five-year survival rate of just 13%. The incidence of pancreatic cancer is also on the rise, making the need for new and effective treatments more urgent than ever. The approval of Optune Pax is a testament to the power of innovation and a symbol of hope for the thousands of patients and families affected by this devastating disease.
The Future of Cancer Care: Non-Invasive, Home-Based, and Patient-Centered
The arrival of Optune Pax signals a broader shift in the future of cancer care. The move towards non-invasive, home-based therapies that prioritize patient quality of life is a trend that is likely to continue. By empowering patients to take a more active role in their treatment and to receive care in the comfort of their own homes, we can create a more humane and effective healthcare system. The success of Optune Pax in treating pancreatic cancer also opens the door for the application of TTFields technology to other solid tumors, offering the potential for even more breakthroughs in the years to come.
For now, the approval of Optune Pax is a moment to celebrate. It is a victory for science, for innovation, and most importantly, for the patients who have been waiting for a new reason to hope. The fight against pancreatic cancer is far from over, but with the arrival of this groundbreaking new therapy, we have a powerful new weapon in our arsenal.
